Healthcare consumers increasingly demand control over their medical decisions, including the ability to order laboratory tests without a physician's prescription. This shift has accelerated the growth of direct-access lab testing, a service model that empowers individuals to request specific blood work, diagnostic panels, and screening tests independently. However, the availability and regulations governing this service vary dramatically across the United States, creating a complex landscape that requires careful navigation.
Understanding Direct-Access Lab Testing Regulations
Direct-access lab testing operates within a framework of state-specific regulations that determine which tests consumers can order independently and under what circumstances. Unlike traditional laboratory services that require physician orders, this model allows individuals to select tests directly, though regulatory requirements vary by jurisdiction.
The Federal and State Regulatory Framework
The Clinical Laboratory Improvement Amendments (CLIA) establish baseline federal standards for laboratory testing quality and accuracy. However, individual states maintain authority over which tests require physician authorization. This creates a patchwork of regulations across the country.
Key regulatory considerations include:
- Scope of practice laws for laboratory personnel
- Required physician oversight levels
- Prohibited test categories
- Consumer notification requirements
- Result reporting obligations
Some states embrace consumer autonomy by permitting most non-prescription laboratory services, while others maintain strict physician-order requirements for all but the most basic tests. Understanding these distinctions is essential for anyone seeking lab testing without insurance or traditional medical gatekeepers.
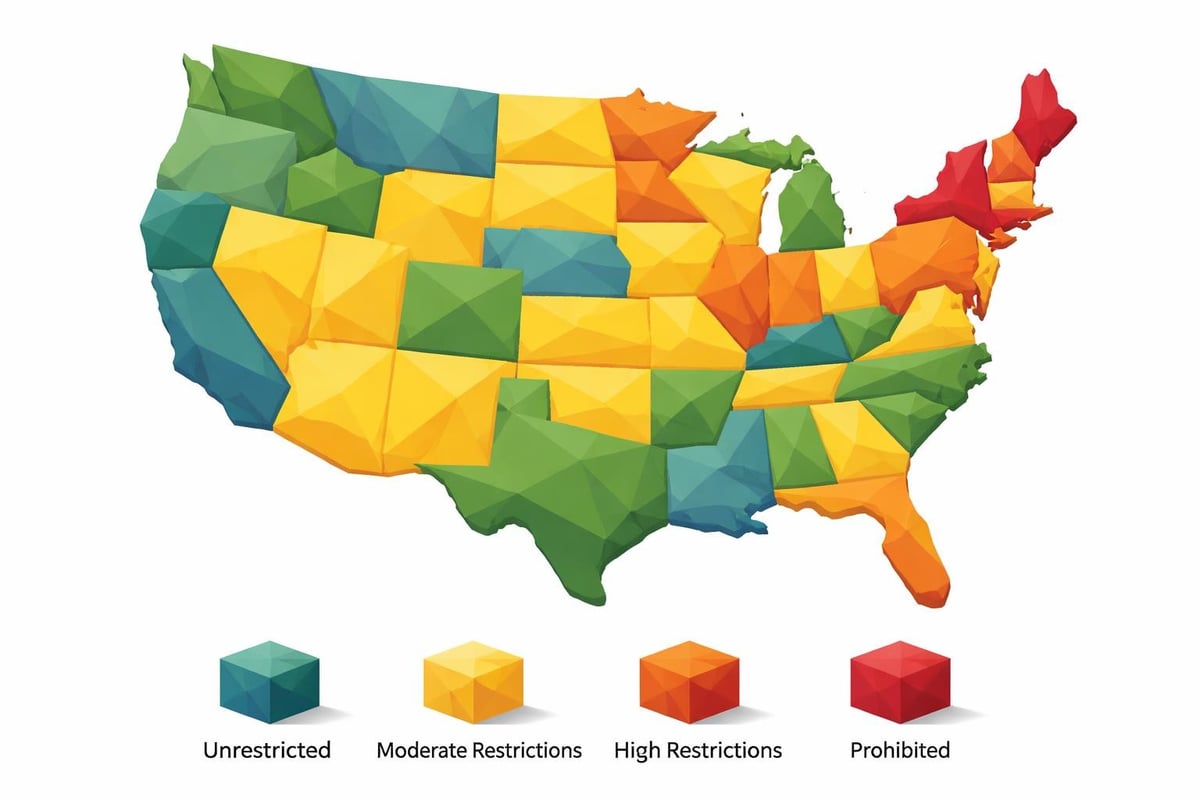
States With Full Direct-Access Lab Testing
Several states have adopted comprehensive policies that support consumer-initiated laboratory services with minimal restrictions. These jurisdictions recognize that informed individuals should have the right to access diagnostic information about their own health.
Unrestricted Access States
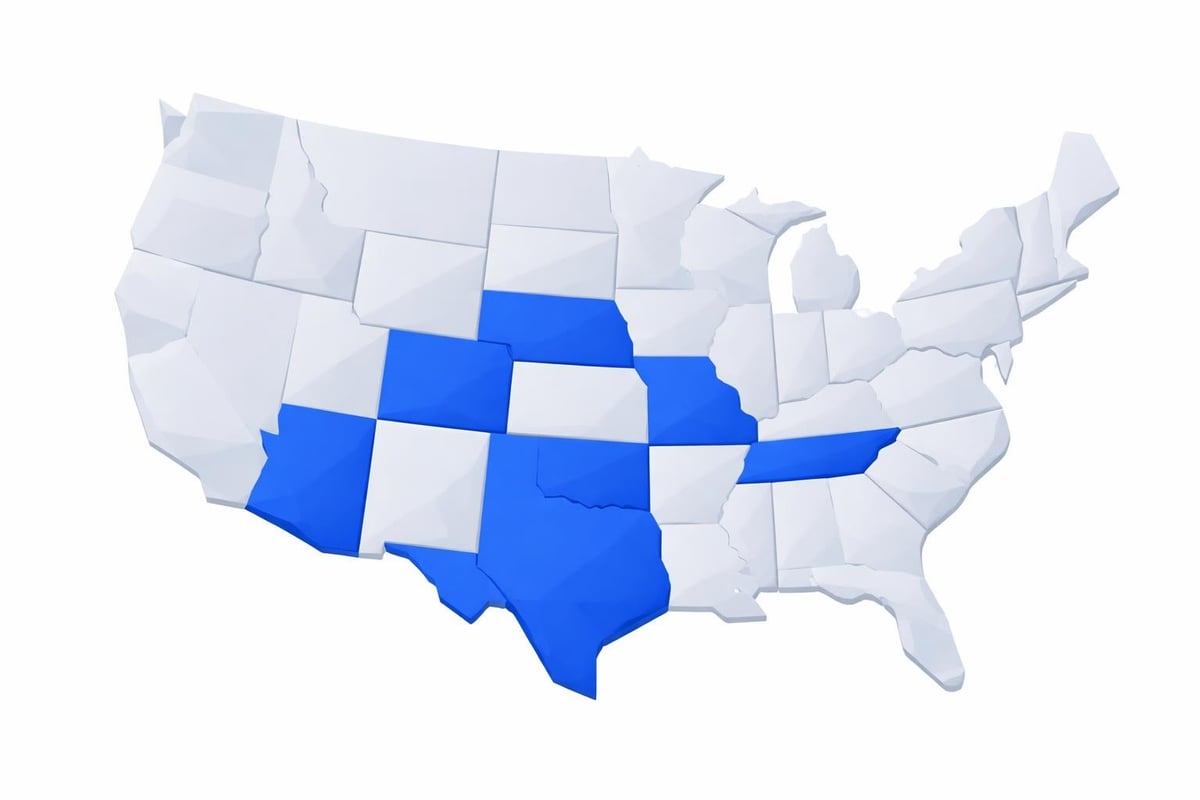
Arizona leads the nation in consumer laboratory access, permitting individuals to order virtually any clinical test without physician involvement. The state's progressive approach reflects a broader commitment to healthcare transparency and patient autonomy.
| State | Restriction Level | Notable Exceptions | Verification Required |
|---|---|---|---|
| Arizona | Minimal | None | No |
| Arkansas | Minimal | Controlled substances | No |
| Colorado | Low | Genetic testing guidelines | Minimal |
| Kansas | Low | None | No |
| Tennessee | Low | None | No |
Colorado follows a similar model, though healthcare providers emphasize compliance with genetic testing guidelines when applicable. Arkansas permits most tests but maintains restrictions on panels related to controlled substance monitoring.
Benefits in Unrestricted States
Consumers in these states enjoy maximum flexibility when managing their health. They can proactively monitor chronic conditions, track wellness markers, or investigate concerning symptoms without the time and expense of medical appointments.
Advantages include:
- Immediate test ordering capability
- Lower overall costs by eliminating consultation fees
- Faster access to results
- Greater privacy in sensitive health matters
- Ability to comparison shop among providers
The convenience factor cannot be overstated. Rather than scheduling appointments weeks in advance, individuals can order labs without a doctor visit and receive results within days.
States With Moderate Restrictions
A larger group of states permits direct-access lab testing but imposes specific limitations on test categories or requires certain administrative procedures. These regulations balance consumer access with professional oversight concerns.
Common Restriction Categories
Most moderately restrictive states prohibit independent ordering of tests that require complex interpretation or carry significant medical implications. Genetic testing, certain cancer markers, and specialized infectious disease panels often fall into restricted categories.
California represents a middle-ground approach. While the state permits many common tests, it requires laboratory directors to review orders and may restrict access to specialized panels. This oversight ensures that consumers receive appropriate guidance about result interpretation.
States with moderate restrictions typically allow:
- Routine blood counts and chemistry panels
- Lipid and metabolic screening
- Thyroid function tests
- Vitamin and mineral deficiency testing
- Common infectious disease screening
Florida, Texas, and Illinois each maintain distinct regulatory frameworks that permit substantial consumer access while preserving some professional gatekeeping. Understanding lab test pricing becomes particularly important in these states, where cost structures may vary based on ordering procedures.
Navigation Strategies
Consumers in moderately restrictive states should research specific test availability before attempting to order. Many laboratories maintain detailed lists of permitted tests, and comprehensive test menus help individuals identify which panels they can access independently.
| State Category | Available Tests | Physician Review | Typical Turnaround |
|---|---|---|---|
| Moderate restriction | 60-80% of common tests | Sometimes required | 1-3 days |
| High restriction | 30-50% of common tests | Usually required | 2-5 days |
| Full restriction | Very limited | Always required | 3-7 days |
Highly Restrictive and Prohibited States
Several states maintain stringent limitations on consumer-initiated laboratory testing, reflecting different philosophical approaches to medical oversight and patient safety. These jurisdictions generally require physician orders for most diagnostic tests.
States Requiring Physician Orders
New York stands as the most restrictive state, essentially prohibiting direct-access lab testing for most clinical purposes. The state's Department of Health maintains that physician oversight protects consumers from misinterpreting results or pursuing inappropriate follow-up care.
New Jersey, Rhode Island, and Maryland similarly restrict independent test ordering. These states argue that the physician-patient relationship ensures proper test selection, accurate interpretation, and appropriate clinical follow-up.
Challenges in restrictive states include:
- Mandatory doctor visits adding time and cost
- Limited consumer autonomy in health decisions
- Reduced access for uninsured or underinsured individuals
- Barriers to preventive health monitoring
- Delayed diagnosis when symptoms are subtle
Despite these restrictions, residents can still access some testing options. Wellness panels, certain screening tests, and basic metabolic markers may remain available through specific programs or facilities that optimize information management systems for compliance.
Working Within Restrictive Frameworks
Even in states with significant limitations, consumers have options. Some laboratories offer physician review services bundled with test orders, providing the required oversight while maintaining convenience and affordability.
Telemedicine platforms have emerged as valuable tools in restrictive states, connecting consumers with physicians who can authorize tests remotely. This hybrid approach satisfies regulatory requirements while preserving many benefits of direct-access models.
Cost Considerations Across State Lines
Pricing for laboratory services varies not only by test type but also by state regulatory environment. Understanding these cost dynamics helps consumers make informed decisions about where to get lab tests and how to budget for health monitoring.
Price Variation Factors
States with fewer restrictions typically see more competitive pricing as multiple providers compete for consumer business. The transparency required in direct-access models also drives prices down compared to traditional insurance-based billing.
Factors influencing costs include:
- State regulatory compliance requirements
- Physician review fees where mandated
- Market competition levels
- Laboratory operational costs
- Insurance participation status
Self-pay options often provide the best value, particularly for routine tests. An estradiol test cost comparison across states reveals significant variation, with direct-access models in unrestricted states typically offering the lowest prices.
Insurance vs. Self-Pay Dynamics
Traditional insurance-based testing often involves copays, deductibles, and surprise billing. Direct-access lab testing circumvents these complexities by offering transparent, upfront pricing that consumers can evaluate before committing.
| Payment Method | Average Cost Range | Price Transparency | Out-of-Pocket Predictability |
|---|---|---|---|
| Insurance-based | $50-$500+ | Low | Low |
| Direct-access self-pay | $20-$200 | High | High |
| Insurance + direct-access | $30-$250 | Moderate | Moderate |
Many consumers discover that paying directly costs less than their insurance copay for the same test. This counterintuitive reality reflects the administrative overhead built into insurance-based medical billing systems.
Special Testing Categories and Restrictions
Certain test types face additional scrutiny regardless of state direct-access policies. Understanding these special categories helps consumers set realistic expectations about what they can order independently.
Genetic and Molecular Testing
Genetic tests present unique interpretive challenges that most states address through additional oversight requirements. Even in permissive jurisdictions, laboratories often require genetic counseling or physician consultation before processing hereditary disease panels.
Direct-to-consumer genetic testing companies operate under different regulatory frameworks than clinical laboratories, creating confusion about which services require medical oversight. Clinical diagnostic genetic tests generally face stricter requirements than ancestry or wellness genetic panels.
Controlled Substance Monitoring
Tests designed to detect drugs of abuse or monitor controlled substance compliance almost universally require physician orders. These restrictions address concerns about test manipulation, result falsification, and inappropriate use of medical information.
Infectious Disease Testing
HIV testing occupies a special category in many states. While rapid HIV tests are widely available without physician orders, confirmatory testing and certain other infectious disease panels may require medical authorization.
Comprehensive testing services typically outline which infectious disease tests fall under direct-access provisions and which require traditional ordering procedures.
FSA and HSA Compatibility
Health Savings Accounts (HSA) and Flexible Spending Accounts (FSA) provide tax-advantaged ways to pay for medical expenses, including laboratory testing. Direct-access lab testing qualifies as an eligible expense under most account terms.
Using Tax-Advantaged Accounts
Consumers can typically use HSA or FSA funds to pay for qualifying laboratory tests without requiring insurance involvement. This creates opportunities for significant savings by combining pretax dollars with competitive direct-access pricing.
Steps to use HSA/FSA funds:
- Verify test eligibility with account administrator
- Order tests through direct-access provider
- Pay using HSA/FSA debit card or request reimbursement
- Retain receipts and documentation for tax records
- Submit claims if reimbursement is required
FSA HSA eligible tests encompass most diagnostic and screening panels that serve medical purposes. Wellness tests without specific diagnostic intent may face additional scrutiny from account administrators.
Result Interpretation and Follow-Up
Accessing tests independently creates responsibility for understanding results and pursuing appropriate follow-up care. This represents both an opportunity and a challenge for consumers embracing direct-access models.
Understanding Laboratory Reports
Modern laboratory reports include reference ranges, flagged abnormal values, and sometimes interpretive comments. However, these standardized elements cannot replace personalized medical advice about what results mean for individual health circumstances.
Understanding lab results requires familiarity with medical terminology, normal ranges, and the clinical significance of deviations. Many direct-access providers offer educational resources to support consumer interpretation.
When to Seek Medical Consultation
Critical or significantly abnormal results warrant immediate medical attention. Direct-access testing empowers proactive health monitoring but should not replace appropriate medical care when results indicate serious conditions.
Consumers should establish relationships with healthcare providers who can interpret results, recommend follow-up testing, and coordinate treatment when necessary. The goal of direct-access testing is informed self-advocacy, not complete medical independence.
Future Trends in Direct-Access Testing
The direct-access lab testing landscape continues evolving as technology advances, consumer expectations shift, and evidence accumulates about the model's safety and effectiveness. Several trends are shaping the industry's trajectory.
Regulatory Harmonization Efforts
Industry advocates are pushing for greater consistency in state regulations, arguing that the current patchwork creates unnecessary barriers to healthcare access. While complete harmonization seems unlikely given states' traditional authority over medical practice, incremental alignment may occur.
Professional organizations are developing best practices for direct-access models that balance consumer autonomy with appropriate safeguards. Healthcare facilities refining their protocols demonstrate how professional standards can support both access and quality.
Technology Integration
Advanced platforms are improving test ordering interfaces, result delivery systems, and interpretive support tools. Artificial intelligence may eventually provide personalized insights that help consumers understand results in the context of their individual health profiles.
Mobile health integration allows consumers to track laboratory results over time, identifying trends that might escape notice in isolated test reviews. This longitudinal perspective adds value beyond what single test instances provide.
Expanding Test Menus
As regulations evolve and laboratory capabilities advance, the range of available direct-access tests continues expanding. Comprehensive test catalogs now include specialized panels that were previously available only through physician orders.
Point-of-care testing technology may further democratize access, enabling some tests to be performed outside traditional laboratory settings while maintaining accuracy and reliability.
Choosing a Direct-Access Provider
Selecting the right direct-access lab testing service requires evaluating multiple factors beyond simple price comparisons. Quality, convenience, and support services all contribute to overall value.
Evaluation Criteria
Consumers should assess provider credentials, laboratory certifications, result turnaround times, and customer support availability. CLIA certification and state licensure represent baseline quality indicators that all legitimate providers must maintain.
Important selection factors:
- Test menu comprehensiveness
- Geographic accessibility of collection sites
- Digital platform usability
- Result delivery methods
- Customer service responsiveness
- Educational resource availability
Finding nearby lab locations ensures convenient specimen collection, reducing the logistical burden of testing. Proximity matters particularly for tests requiring fasting or time-sensitive collection.
Transparency and Communication
Reputable providers clearly communicate pricing, turnaround times, and any limitations before consumers commit to testing. Hidden fees, unclear processes, or vague result reporting timelines signal potential problems.
Detailed service explanations help consumers understand what to expect at each step, from ordering through result receipt. This transparency builds trust and ensures informed decision-making.
State-Specific Resources and Information
Each state with direct-access provisions maintains resources to help consumers navigate available services. State health departments, professional laboratory associations, and individual testing facilities provide guidance tailored to local regulations.
Accessing State Information
State health department websites typically outline direct-access testing regulations, permitted test categories, and compliance requirements. These official resources provide authoritative information about what consumers can legally access.
Professional laboratory organizations maintain directories of certified facilities offering direct-access services. These listings help consumers identify reputable providers operating within regulatory frameworks.
Local Laboratory Options
Regional and local laboratories often provide more personalized service than national chains, though they may offer smaller test menus. Community-based testing facilities frequently combine direct-access convenience with relationships that support ongoing health monitoring.
National providers offer consistency across locations, standardized processes, and often more competitive pricing through economies of scale. The choice between local and national services depends on individual priorities and specific testing needs.
Direct-access lab testing represents a significant shift toward consumer empowerment in healthcare, though availability depends heavily on state regulations and individual circumstances. Understanding the regulatory landscape, cost structures, and quality considerations enables informed decisions about independent health monitoring. Justlabs simplifies this process by offering transparent pricing, quick results, and convenient access to comprehensive testing without the complexity of insurance billing or mandatory doctor visits.